While we try to keep things accurate, this content is part of an ongoing experiment and may not always be reliable.
Please double-check important details — we’re not responsible for how the information is used.
Biochemistry Research
“Cutting to Survive: Unraveling the Molecular Mechanism of DNA Bridge Resolution in Cell Division”
Scientists have elucidated the molecular mechanism by which LEM-3 cuts DNA bridges during cytokinesis.
Biochemistry Research
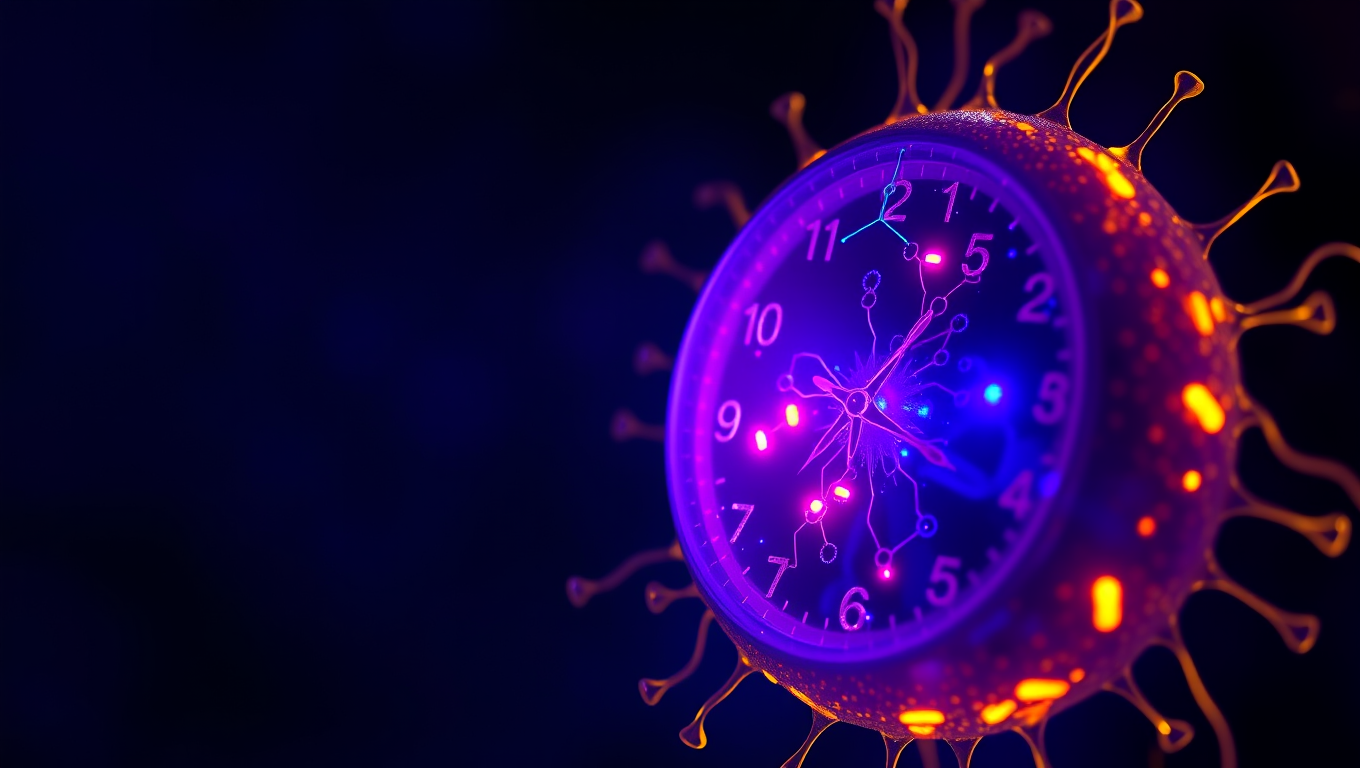
“Unlocking Timekeeping Secrets: Scientists Reveal How Artificial Cells Can Accurately Keep Rhythm”
Scientists at UC Merced have engineered artificial cells that can keep perfect time—mimicking the 24-hour biological clocks found in living organisms. By reconstructing circadian machinery inside tiny vesicles, the researchers showed that even simplified synthetic systems can glow with a daily rhythm—if they have enough of the right proteins.
Behavioral Science
The Sugar that Sparked Life: Unraveling the Mystery of Ribose’s Preeminence in RNA Development
What made ribose the sugar of choice for life’s code? Scientists at Scripps Research may have cracked a major part of this mystery. Their experiments show that ribose binds more readily and selectively to phosphate compared to other similar sugars, forming a structure ideal for RNA formation. This discovery hints at how nature might have selected specific molecules long before enzymes or life existed, and could reshape our understanding of life’s chemical origins.
Biochemistry Research
The Whispering Womb: Uncovering the Secret Language of Embryonic Cells
Scientists found that embryonic skin cells “whisper” through faint mechanical tugs, using the same force-sensing proteins that make our ears ultrasensitive. By syncing these micro-movements, the cells choreograph the embryo’s shape, a dance captured with AI-powered imaging and computer models. Blocking the cells’ ability to feel the whispers stalls development, hinting that life’s first instructions are mechanical. The discovery suggests hearing hijacked an ancient force-sensing toolkit originally meant for building bodies.
-

 Detectors11 months ago
Detectors11 months agoA New Horizon for Vision: How Gold Nanoparticles May Restore People’s Sight
-

 Earth & Climate12 months ago
Earth & Climate12 months agoRetiring Abroad Can Be Lonely Business
-

 Cancer12 months ago
Cancer12 months agoRevolutionizing Quantum Communication: Direct Connections Between Multiple Processors
-

 Albert Einstein12 months ago
Albert Einstein12 months agoHarnessing Water Waves: A Breakthrough in Controlling Floating Objects
-

 Chemistry12 months ago
Chemistry12 months ago“Unveiling Hidden Patterns: A New Twist on Interference Phenomena”
-

 Earth & Climate12 months ago
Earth & Climate12 months agoHousehold Electricity Three Times More Expensive Than Upcoming ‘Eco-Friendly’ Aviation E-Fuels, Study Reveals
-

 Diseases and Conditions12 months ago
Diseases and Conditions12 months agoReducing Falls Among Elderly Women with Polypharmacy through Exercise Intervention
-

 Agriculture and Food12 months ago
Agriculture and Food12 months ago“A Sustainable Solution: Researchers Create Hybrid Cheese with 25% Pea Protein”