While we try to keep things accurate, this content is part of an ongoing experiment and may not always be reliable.
Please double-check important details — we’re not responsible for how the information is used.
Agriculture and Food
Unlocking Duckweed’s Secrets: A Plant that Farms Itself and Holds Key to Sustainable Future
Scientists have released new and more accurate genome sequences for five species of duckweed. Their research reveals the specific genes responsible for some of the plant’s most useful traits, allowing for new commercial agriculture applications.

Agriculture and Food
Unearthing Life’s Secrets: Deep Microbes Thrive without Sunlight
Chinese scientists uncovered a powerful energy source for deep Earth microbes: hydrogen and oxidants generated by rock fracturing during earthquakes. The process may also suggest how life could exist on other planets without sunlight.
Agriculture and Food
Breaking New Ground: Scientists Develop Groundbreaking Chromosome Editing Technology
A group of Chinese scientists has created powerful new tools that allow them to edit large chunks of DNA with incredible accuracy—and without leaving any trace. Using a mix of advanced protein design, AI, and clever genetic tweaks, they’ve overcome major limitations in older gene editing methods. These tools can flip, remove, or insert massive pieces of genetic code in both plants and animals. To prove it works, they engineered rice that’s resistant to herbicides by flipping a huge section of its DNA—something that was nearly impossible before.
Agriculture and Food
The Ancient Origins of Potatoes Revealed
About 9 million years ago, a wild interspecies fling between tomato-like plants and potato relatives in South America gave rise to one of the world’s most important crops: the potato. Scientists have now traced its roots to a rare natural hybridization that created the tuber, a storage organ that allowed the plant to survive harsh Andean environments and spread rapidly.
-

 Detectors1 year ago
Detectors1 year agoA New Horizon for Vision: How Gold Nanoparticles May Restore People’s Sight
-

 Cancer1 year ago
Cancer1 year agoRevolutionizing Quantum Communication: Direct Connections Between Multiple Processors
-

 Earth & Climate1 year ago
Earth & Climate1 year agoRetiring Abroad Can Be Lonely Business
-

 Albert Einstein1 year ago
Albert Einstein1 year agoHarnessing Water Waves: A Breakthrough in Controlling Floating Objects
-

 Chemistry1 year ago
Chemistry1 year ago“Unveiling Hidden Patterns: A New Twist on Interference Phenomena”
-

 Earth & Climate1 year ago
Earth & Climate1 year agoHousehold Electricity Three Times More Expensive Than Upcoming ‘Eco-Friendly’ Aviation E-Fuels, Study Reveals
-

 Diseases and Conditions1 year ago
Diseases and Conditions1 year agoReducing Falls Among Elderly Women with Polypharmacy through Exercise Intervention
-

 Earth & Climate1 year ago
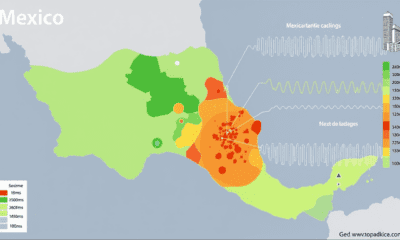
Earth & Climate1 year agoPredicting Damage from Local Earthquakes in Mexico City