While we try to keep things accurate, this content is part of an ongoing experiment and may not always be reliable.
Please double-check important details — we’re not responsible for how the information is used.
Colon Cancer
The Key to Unlocking Better Leukemia Treatments: A New Understanding of Gene Mutations and Cell Maturity
An international study has uncovered why a widely used treatment for acute myeloid leukemia (AML) doesn’t work for everyone. The findings could help doctors better match patients with the therapies most likely to work for them.
Colon Cancer
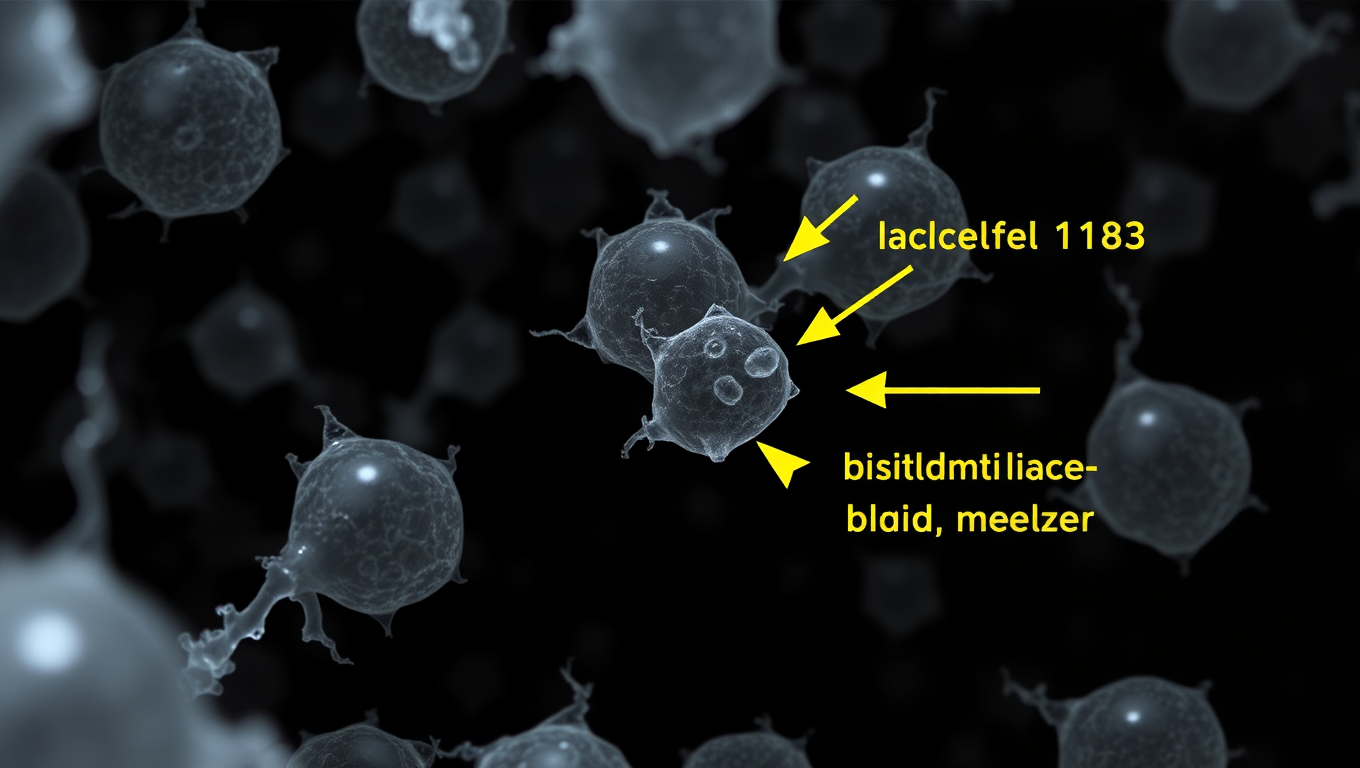
Scientists Discover a Tiny Molecule That Could Revolutionize Weight Loss Treatment
Researchers at the Salk Institute have used CRISPR to uncover hidden microproteins that control fat cell growth and lipid storage, identifying one confirmed target, Adipocyte-smORF-1183. This breakthrough could lead to more effective obesity treatments, surpassing the limitations of current drugs like GLP-1.
Breast Cancer
Reversing Alzheimer’s Damage: A Surprising Breakthrough with Cancer Drugs
In an exciting breakthrough, researchers have identified cancer drugs that might reverse the effects of Alzheimer’s disease in the brain. By analyzing gene expression in brain cells, they discovered that some FDA-approved cancer medications could reverse damage caused by Alzheimer’s.
Cholesterol
The Pistachio Paradox: How Swapping Bedtime Snacks Can Rewire Your Gut and Help Prevent Diabetes
A new study reveals that swapping a typical nighttime carbohydrate snack for pistachios may beneficially alter gut bacteria in people with prediabetes. Conducted by Penn State researchers, the 12-week clinical trial found that pistachio consumption increased beneficial gut microbes like Roseburia and reduced harmful ones such as Blautia hydrogenotrophica. These microbiome changes could potentially support metabolic health and slow the progression to Type 2 diabetes. While more research is needed to confirm health outcomes, this study positions pistachios as a promising late-night snack with microbiome-boosting potential.
-

 Detectors1 year ago
Detectors1 year agoA New Horizon for Vision: How Gold Nanoparticles May Restore People’s Sight
-

 Cancer1 year ago
Cancer1 year agoRevolutionizing Quantum Communication: Direct Connections Between Multiple Processors
-

 Earth & Climate1 year ago
Earth & Climate1 year agoRetiring Abroad Can Be Lonely Business
-

 Albert Einstein1 year ago
Albert Einstein1 year agoHarnessing Water Waves: A Breakthrough in Controlling Floating Objects
-

 Earth & Climate1 year ago
Earth & Climate1 year agoHousehold Electricity Three Times More Expensive Than Upcoming ‘Eco-Friendly’ Aviation E-Fuels, Study Reveals
-

 Chemistry1 year ago
Chemistry1 year ago“Unveiling Hidden Patterns: A New Twist on Interference Phenomena”
-

 Diseases and Conditions1 year ago
Diseases and Conditions1 year agoReducing Falls Among Elderly Women with Polypharmacy through Exercise Intervention
-

 Agriculture and Food1 year ago
Agriculture and Food1 year ago“A Sustainable Solution: Researchers Create Hybrid Cheese with 25% Pea Protein”